Silanes, siloxanes, or mixtures thereof are commonly used hydrophobic impregnating agents.
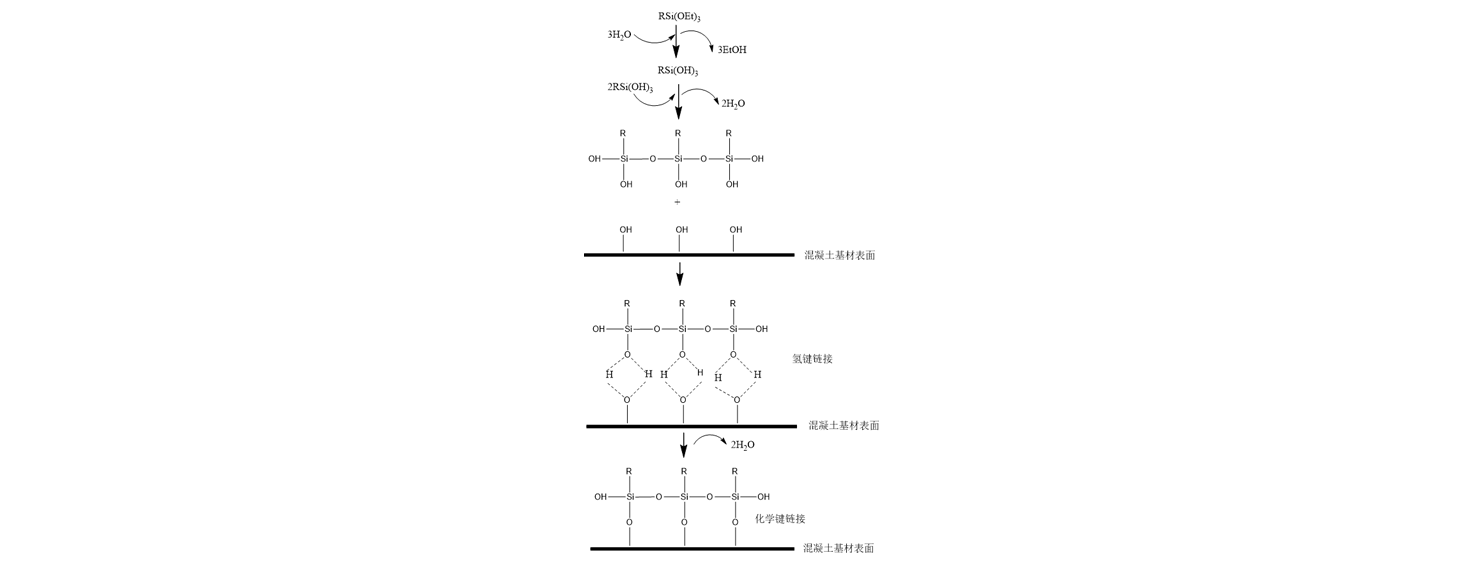
The permeability of silanes and siloxanes is determined by their small-molecule structures; silanes have a molecular diameter of approximately 1.0 × 10⁻⁶ to 1.5 × 10⁻⁶ nm, while siloxanes have a molecular diameter of approximately 1.5 × 10⁻⁶ to 7.5 × 10⁻⁶ nm. Both silanes and siloxanes contain alkyl and alkoxy groups. The alkyl groups serve to lower the surface tension of the concrete and enhance its hydrophobic properties, while the alkoxy groups undergo hydrolysis to form silanols. These silanols can bond with hydroxyl-containing compounds within the concrete via hydrogen bonds, or—within the alkaline environment of the concrete—they can undergo condensation to form silicates, thereby establishing stronger chemical bonds. The small-molecule structure of silane materials dictates their excellent permeability; they can penetrate to a depth of 3–20 mm beneath the surface of a concrete substrate. The actual depth of penetration is influenced by factors such as the application rate, substrate strength, concrete water-binder ratio, concrete moisture content, and the specific type of silane used. The mechanism by which silanes bond with concrete is illustrated in Figure 2. Silane-based materials constitute a class of substances characterized by the molecular structure depicted in Figure 1. Structurally, a silane molecule can be divided into two distinct parts: a protective group (R) and a bonding group [Si(OR’)₃]. Under alkaline conditions, the Si(OR’)₃ group undergoes hydrolysis with the elimination of alcohol to form a silanetriol. This silanetriol exhibits a strong affinity for inorganic substrates, bonding to them via either hydrogen bonds or covalent bonds. Simultaneously, the non-polar organic groups (R) orient themselves outward to form a hydrophobic layer; this process creates a water-repellent surface on the concrete, thereby enhancing its overall durability. Based on the aforementioned analysis of the chemical structure of silane materials, the following conclusions can be drawn: (1) A chemical reaction between the silane and the concrete substrate is a prerequisite for the silane to effectively provide protective benefits to the concrete; and (2) while different silane materials react with concrete according to the same fundamental principle, they differ in the specific hydrophobic efficacy provided by their respective hydrophobic functional groups.
Factors Influencing Penetration Depth: The primary factors determining the penetration depth of silane materials depend mainly on the quantity of silane applied and the duration of the penetration process. Specifically, the influence of silane materials on penetration depth can be categorized into the following aspects: 1. The Impact of Application Rate on Penetration Depth: The quantity of silane material applied directly affects its penetration depth within the concrete matrix. Under identical conditions, a higher application rate of silane results in a greater penetration depth, potentially reaching a maximum of 50 mm. When the application rate remains constant, however, variations in concrete strength (or density) will influence the silane’s penetration rate; specifically, higher concrete strength corresponds to a slower silane penetration rate. Therefore, when assessing the penetration depth in concrete, it is essential to account for the specific concrete strength (or density) to ensure that sufficient time is allowed for complete penetration; failure to do so may compromise the accuracy of the test results. Consequently, for effective silane-based corrosion protection, the application rate must be tailored to specific project requirements to achieve the desired penetration depth and protective efficacy. 2. Loss of Active Ingredients Due to Volatilization: As silane is a low-molecular-weight substance, it is highly susceptible to volatilization prior to reacting with the concrete—particularly under windy or high-temperature conditions. Once the silane material has undergone a chemical reaction with the concrete, however, it becomes an integral part of the concrete structure and is no longer subject to volatilization. During winter construction, when ambient temperatures are low, the reaction rate between the silane and the concrete decreases. According to physicochemical principles, for every 10°C drop in temperature, the rate of chemical reaction slows down by a factor of 2 to 4. Consequently, any silane that does not have sufficient time to react with the concrete is prone to volatilization, thereby reducing the effective quantity of silane available for protection.
Impact on Hydrophobic Performance: The hydrophobic effect is determined by the portion of the silane molecule that does not undergo chemical reactions with the concrete substrate. Currently, common hydrophobic functional groups found on the market include methyl, propyl, isobutyl, n-octyl, and isooctyl groups. These distinct hydrophobic functional groups not only influence the degree of hydrophobicity but also affect the concrete’s water absorption rate and its resistance to chloride ion ingress. 1. Influence of Silane Chemical Composition: Methyl and propyl groups, possessing hydrophobic functional groups with only one or three carbon atoms respectively, can generate a “beading effect” (water droplet formation) on the concrete surface for a short duration; however, their hydrophobic efficacy diminishes after prolonged immersion in water, rendering them unsuitable for long-term concrete corrosion protection. Although the isobutyl group contains a hydrophobic functional group with only four carbon atoms, its unique molecular structure endows it with not only excellent hydrophobic properties but also superior resistance to chloride ion ingress. Both n-octyl and isooctyl groups feature hydrophobic moieties comprising eight carbon atoms; consequently, they exhibit enhanced hydrophobic performance and superior resistance to chloride ion ingress. Furthermore, due to the structural similarities between isooctyl and isobutyl groups—specifically their unique branching structures—the protective efficacy of the isooctyl group surpasses that of the n-octyl group. As the number of carbon atoms within the hydrophobic functional group increases, the silane material’s hydrophobic performance and resistance to chloride ion ingress generally improve; however, longer carbon chains tend to compromise the material’s penetration depth, thereby limiting its practical application in concrete corrosion protection. 2. Influence of Superhydrophobic Agents in Silane Formulations: Incorporating superhydrophobic agents into silane formulations can significantly enhance the silane’s hydrophobic efficacy. This is particularly beneficial in environments where the concrete surface is likely to be exposed to standing water shortly after silane application (e.g., tidal zones or regions with frequent precipitation); in such scenarios, the addition of a superhydrophobic agent helps prevent the premature loss of the silane material caused by contact with standing water.
